
XENOVIEW® Provides Effective Measurement of Regional Lung Ventilation
Study Design
- Two prospective, multicenter, randomized, open-label, crossover clinical studies that compared XENOVIEW® magnetic resonance imaging (MRI) to xenon Xe 133 scintigraphy in adult patients with pulmonary disorders:
‒ Study 1: patients being evaluated for possible lung resection surgery (n = 32)*
‒ Study 2: patients being evaluated for possible lung transplant surgery (n = 49)† - Primary analysis: pre-specified equivalence interval between XENOVIEW® and xenon Xe 133 imaging
- Patient population with a broad representation of underlying pulmonary diseases
- Six-zone analysis of regional lung ventilation
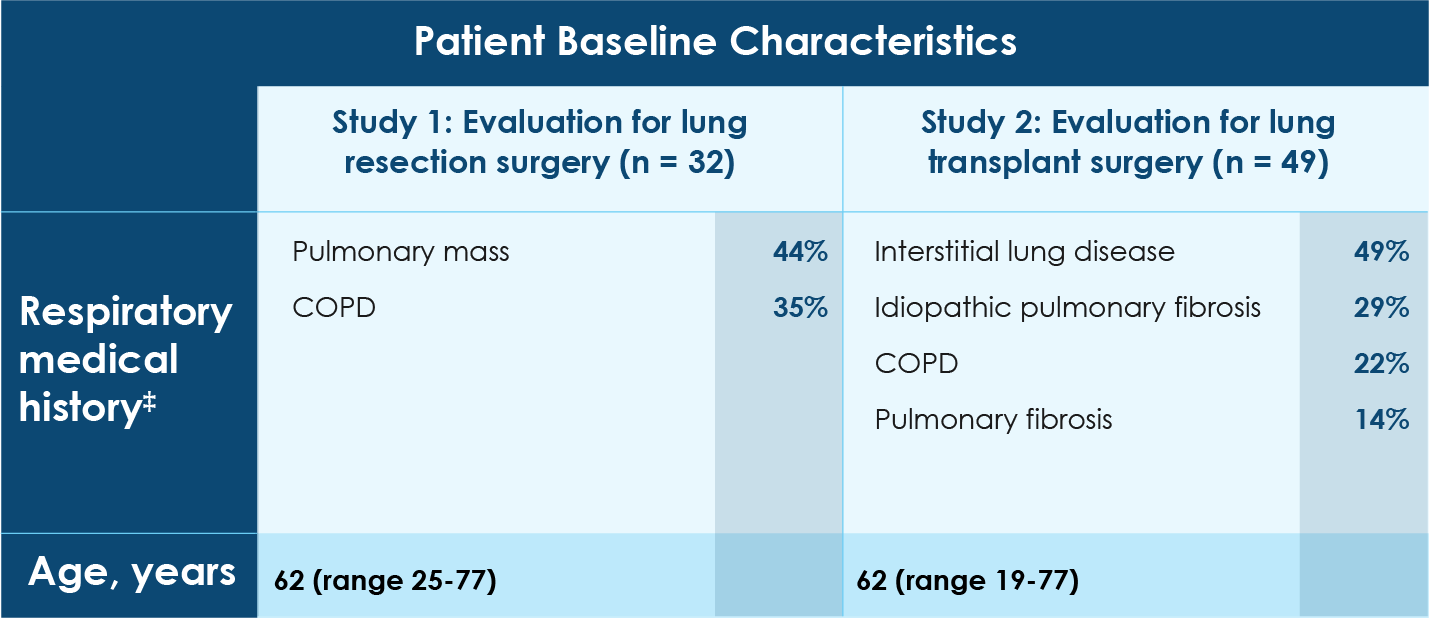
*On each XENOVIEW® and xenon Xe 133 scan, the fraction of total signal in the lungs was calculated in each of 6 zones consisting of upper, middle, and lower regions in each lung. These values were used to estimate the postoperative percentage of lung ventilation predicted to remain after planned resection of a pre-specified lung area.
†On each XENOVIEW® and xenon Xe 133 scan, right lung signal and total signal in the lungs were calculated and used to estimate the percentage of overall lung ventilation contributed by the right lung.
‡Medical history may not be the primary reason for lung resection or lung transplant surgery.
Study Results
XENOVIEW®

Xenon Xe 133 Scintigraphy


*One of the 32 subjects who completed both scans was excluded from the primary analysis because no planned resection area was recorded.
XENOVIEW® Was Well Tolerated
- Adverse reactions were reported in 14% (12/83) of patients (98% of whom received a single administration of XENOVIEW®)
- The only adverse reactions reported by more than 1 patient were oropharyngeal pain (n = 4), headache (n = 2), and dizziness (n = 2)
